 Eight individual protein subunits, named either α to θ or 1 to 8 (all products of essential genes in yeast) assemble to form a double-ringed barrel structure ( Figure 1), which is required for the folding of newly synthesized actin and tubulin molecules and a somewhat restricted range of other folding substrates. The Molecular Chaperone CCTĬCT is a member of the chaperonin family of molecular chaperones and is found in all eukaryotes. 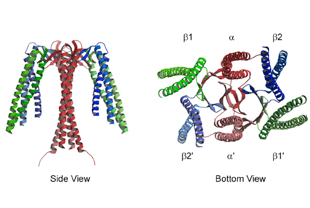 
The focus of this review is the ATP-dependent chaperone chaperonin containing tailless complex polypeptide 1 (CCT) also known as tailless complex polypeptide 1 ring complex (TRiC) and a potential role for CCT in the modulation and/or suppression of protein aggregation will be discussed. Conversely, the small Hsp proteins prevent aggregation via ATP-independent mechanisms (for a comprehensive review, see for example, Hartl et al., 2011). For example, the Hsp70 family, together with the co-chaperone Hsp40 act by binding to nascent or unfolded polypeptide chains via stretches of hydrophobic sequences, whilst Hsp90 proteins bind to and stabilize a more limited sub set of proteins, both in an ATP-dependent manner. The chaperones consist of several different families of proteins, which contribute to maintaining proteostasis by different mechanisms. If cells become deficient in their chaperoning capacity during aging or if proteostasis is disrupted as a consequence of protein misfolding diseases, then modulation of chaperone activity may provide a mechanism to combat the formation of potentially toxic protein aggregates. These observations indicate potential complex changes in the chaperoning requirements and chaperoning capacities of cells. (2014), demonstrates that during aging there is a trend where ATP-dependent chaperones are down regulated, whilst ATP-independent chaperones are up regulated. The array of chaperones present in the cell, the “chaperome,” can change during aging and in neurodegenerative disease states and a detailed analysis of this, performed by Brehme et al. In between this “birth and death” of proteins, the molecular chaperones provide assistance to many proteins, ensuring correct conformations are reached/maintained and misfolded proteins can be unfolded to either refold or be degraded. Numerous events contribute to proteostasis, such as transcription/translation and proteolysis, to ensure that protein levels are optimal. The maintenance of proteostasis is paramount to cellular health. Here there will be a focus on how mechanistically CCT may be able to achieve this and if this potential function of CCT provides any insights and directions for developing future treatments for protein aggregation driven neurodegenerative diseases generally, many of which are associated with aging. Since observations were made in worms over a decade ago using an RNAi screen, which connected CCT subunits to the aggregation of polyglutamine tracts, a role for CCT as a potential modulator of protein aggregation has started to emerge. CCT is also involved in the folding of some additional protein substrates and some CCT subunits have been shown to have functions when monomeric. CCT is required for the folding of the abundant cytoskeletal proteins actin and tubulin, which in turn form assemblies of microfilaments and microtubules. Thus a geometrically defined binding interface is formed from the divergent sequences within the CCT subunit substrate binding domains. When assembled, each of the eight CCT subunits occupies a specific position within each chaperonin ring. It is a multi-subunit oligomer of two rings of eight individual protein subunits. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
